Announcement Key Points
- Hera and Demeetra stem from the gene editing platforms company Transposagen (now a Hera BioLabs company) which is best known for its commercialization of the piggyBac® transposase
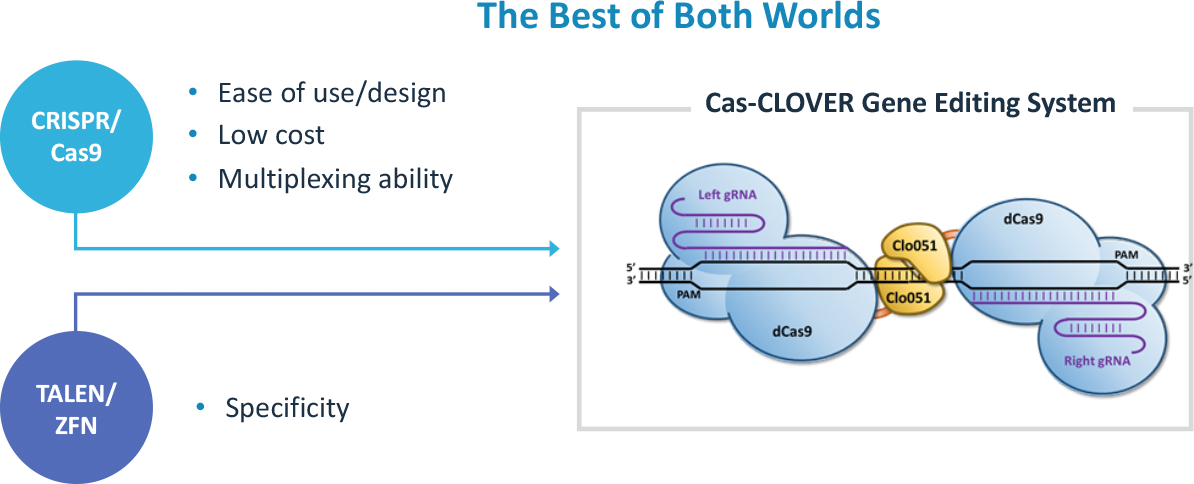
- Cas-CLOVER differs from single-guided CRISPR and dual-guided CRISPR nickase systems as it is truly dimeric and demonstrates high fidelity with no detectable off-targets while maintaining robust editing efficiency
- Issued patents cover these technologies enabling clear freedom-to-operate
- Evaluation opportunities and services for Cas-CLOVER which can convert into full research and commercial licenses with very accessible terms are available
- Demeetra is operating in the pharmaceutical/industrial bioprocessing space as well as agricultural biotechnology, while Hera is focused on products tools and services for research and drug discovery
Targeted nuclease gene editing is a powerful tool for accelerating and improving opportunities for successful drug and cell therapy development from target identification and candidate selection through the production of GMP biologics. And has been used successfully to engineer T-cell therapies. Technologies such as ZFNs, CRISPR/Cas9 and now Cas-CLOVER bind to the target DNA and generate insertions and deletions (“indels”) resulting in “knockout” mutations or targeted “knock-ins” when supplied with the appropriate guide RNA(s) and donor vector.
- CRISPR/Cas9 was a breakthrough for gene editing since it offers a simple and cost-effective method for high efficiency modification of the desired target locus. CRISPR does have a known trade-off as the technology demonstrates high degrees of off target mutagenesis, modifying the genome at undesired locations, in addition to the gene of interest. Off-target mutagenesis also occurs with the dual guided CRISPR nickase system which has been characterized as not truly dimeric (1, 2). Furthermore, licensing restrictions or lack of clear commercial freedom to operate have limited CRISPR/Cas9 use commercially.
Unlike CRISPR/Cas9, which is a monomeric nuclease targeted by a single guide RNA , Cas-CLOVER is a dimeric nuclease system targeted by 2 guide RNAs and has no detectable off-target mutations (3).
The Cas-CLOVER dimeric system uses two guide RNAs targeting “half-site” sequences. The two half-sites are separated by a spacer region where genomic DNA is cleaved with the proprietary dimeric Clo51 nuclease. The stringency of the dimeric Clo51 nuclease gives it the specificity that has shown no off-target activity after millions of next generation sequencing (NGS) reads in human T-cells. Guide RNA design for any gene of interest is flexible as the spacer region between the guides is ideally between 16-30 bases (4).
“High fidelity editing is extremely important to us and our partners as off-targets can effect important characteristics such as growth rate and cloning efficiency in bioprocessing systems” said Jack Crawford CEO of Demeetra. Demeetra and Hera recently collaborated to validate Cas-CLOVER in Chinese hamster ovary (CHO) cell lines for bioprocessing. “Cas-CLOVER is a perfect fit for us as we move into crop trait engineering where we want to minimize off-target risks and reduce timelines due to backcrossing which is required with traditional chemical mutagenesis” he added.
Hera’s SRG OncoRat® is another example of nuclease applications. The Sprague Dawley strain was engineered to contain a knockout in both the Rag2 and IL2rg genes resulting in a fully immunodeficient rat. Dozens of human tumor models have been validated in the OncoRat (link to some examples), including a prostate cancer model recently published in a top tier drug development journal (5).
“Cell and gene therapy research may especially benefit from Cas-CLOVER where off-target mutagenesis is unacceptable” added Mike Schlosser the CEO of Hera. Both proliferating and resting human T-cells have been successfully targeted by Cas-CLOVER to remove cell surface markers associated with graft versus host disease (GVHD) (4).
About Hera
Hera Biolabs, Inc., based in Lexington, KY is an innovative preclinical contract services, products and licensing company. Our mission is to use cutting-edge gene-editing technologies to accelerate preclinical research and drug and cell-based therapeutic discovery and development. Hera built the SRG platform, an immunocompromised double knockout Sprague-Dawley rat engineered for T-cell, B-cell, and NK cell deficiency. Our first established commercial model is the SRG OncoRat, a valuable complement or alternative to oncology studies in mice, and well suited for xenograft oncology studies given its excellent tumor take-rates, large tumor mass, and smooth transition from efficacy to safety. Hera sells the SRG OncoRat to researchers and has been conducting oncology/pharmacology studies for clients over the past 5 years. Using immunocompromised husbandry procedures, our experienced staff performs studies in our on-site state-of-the-art vivarium that features dual HEPA-filtered, disposable IVC rodent caging systems. We’re also continuing to use our proprietary Cas-CLOVER CRISPR system and piggyBac gene-editing technologies and know-how to expand our robust pipeline of models and services.
About Demeetra
Demeetra AgBio is “Engineering Traits for Life” by introducing gene editing technologies in the agriculture, industrial, pharmaceutical & animal health industries. We provide licenses for Cas-CLOVER (the clean CRISPR/Cas9 alternative) and piggyBac transposase or partner under joint development agreements for applications in the engineering of crops and agricultural animals, plant-based drug development and enhanced bioprocessing in human & animal health.
References
1. Tsai et al. (2014) Dimeric CRISPR RNA-guided FokI nucleases for highly specific genome editing. Nature Biotechnology.
2. Lee et al (2015) Site-specific integration in CHO cells mediated by CRISPR/Cas9 and homology-directed DNA repair pathway. Scientific Reports
3. Li et al (2018) Cas-CLOVER™: A High-Fidelity Genome Editing System for Safe and Efficient Modification of Cells for Immunotherapy. Precision CRISPR Congress Poster.
4. Wang et al. (2017) Genome Editing of Therapeutic CAR-T Cells Using A Novel Clo51-dCas9 (NextGEN™) CRISPR System. American Society for Cell & Gene Therapy Poster.
5. Ponnusamy (2019) Orally -Bioavailable Androgen Receptor Degrader, Potential Next-Generation Therapeutic for Enzalutamide-Resistant Prostate Cancer. Clinical Cancer Research























